|
A large contribution to replication fidelity comes from a finely tuned polymerase active site that makes errors in only one out of 10 5 base pairs while incorporating nucleotides at rates exceeding 300 per second ( 1, 2). We also characterize the exonuclease stereospecificity using phosphorothioate-modified DNA, provide a homology model for the DNA primer strand in the exonuclease active site, and propose a dynamic structural model for the transfer of DNA from the polymerase to the exonuclease active site based on MD simulations.ĭNA polymerases have evolved to efficiently copy genomes with extremely high fidelity to fulfill their critical role in maintaining genome stability. 
Because the polymerase stalls after incorporation of a mismatch and after incorporation of one or two correct bases on top of a mismatch, the net contribution of the exonuclease is a function of multiple opportunities to correct mistakes. We show that while proofreading of a terminal mismatch is efficient, proofreading a mismatch buried by one or two correct bases is even more efficient. The contribution of the exonuclease to net fidelity is a function of the kinetic partitioning between extension and excision. Here we characterize the substrate specificity for the proofreading exonuclease of a high-fidelity DNA polymerase by investigating the proofreading kinetics on various DNA substrates. Despite the importance of proofreading to maintaining genome stability, it remains much less studied than the fidelity mechanisms employed at the polymerase active site. While high-fidelity DNA polymerases favor canonical base pairs over mismatches by a factor of ∼1 × 10 5, fidelity is further enhanced several orders of magnitude by a 3′–5′ proofreading exonuclease that selectively removes mispaired bases in the primer strand. The exo-Klenow fragment is used in some fluorescent labeling reactions for microarray, and also in dA and dT tailing, an important step in the process of ligating DNA adapters to DNA fragments, frequently used in preparing DNA libraries for Next-Gen sequencing.Faithful replication of genomic DNA by high-fidelity DNA polymerases is crucial for the survival of most living organisms. This form of the enzyme is called the exo- Klenow fragment. This results in forms of the enzyme being expressed that retain 5' → 3' polymerase activity, but lack any exonuclease activity (5' → 3' or 3' → 5'). This problem can be overcome by introducing mutations in the gene that encodes Klenow. Just as the 5' → 3' exonuclease activity of DNA polymerase I from E.coli can be undesirable, the 3' → 5' exonuclease activity of Klenow fragment can also be undesirable for certain applications. The Klenow fragment was also the original enzyme used for greatly amplifying segments of DNA in the polymerase chain reaction (PCR) process, before being replaced by thermostable enzymes such as Taq polymerase. Filling in receded 3' ends of DNA fragments to make 5' overhang blunt.Synthesis of double-stranded DNA from single-stranded templates. 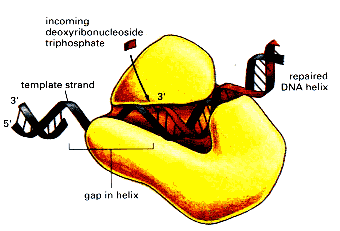
The Klenow fragment is extremely useful for research-based tasks such as: coli makes it unsuitable for many applications, the Klenow fragment, which lacks this activity, can be very useful in research. 5' → 3' polymerase activity, and 3' → 5' exonuclease activity).īecause the 5' → 3' exonuclease activity of DNA polymerase I from E. 
coli is cleaved by subtilisin retains the 5' → 3' exonuclease activity but does not have the other two activities exhibited by the Klenow fragment (i.e. The other smaller fragment formed when DNA polymerase I from E. First reported in 1970, it retains the 5' → 3' polymerase activity and the 3’ → 5’ exonuclease activity for removal of precoding nucleotides and proofreading, but loses its 5' → 3' exonuclease activity. coli is enzymatically cleaved by the protease subtilisin. The Klenow fragment is a large protein fragment produced when DNA polymerase I from E. Functional domains in the Klenow Fragment (left) and DNA Polymerase I (PDB).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
